
Optimize Your Research
Mouse Complement C1Q Tumor Necrosis Factor Related Protein 5, CTRP5 ELISA Kit
| USER INSTRUCTION |
| Cat.No | E1061Mo |
| Standard Curve Range | 70-15000ng/L |
| Sensitivity | 34.23ng/L |
| Size | 48T, 96T |
| Storage | Store at -20°C for one year. Or store at 2-8°C for 6 months. If individual reagents are opened it is recommended that the kit be used within 1 month. Avoid repeated thaw cycles. |
*This product is for research use only, not for use in diagnosis procedures. It’s highly recommended to read this instruction entirely before use.
Precision
Intra-Assay Precision (Precision within an assay) Three samples of known concentration were tested on one plate to assess intra-assay precision. We measured random samples of E1061Mo within the same batch/lot to ensure the consistency of the kits' performances.
| Intra-assay | Sample | n | Mean | Standard deviation | CV (%) |
| Intra-Assay | 1 | 18 | 2320.54 | 111.28 | 4.8 |
| Intra-Assay | 2 | 18 | 5258.94 | 215.06 | 4.09 |
| Intra-Assay | 3 | 18 | 1570.45 | 65.46 | 4.17 |
Inter-Assay Precision (Precision between assays) Three samples of known concentration were tested in separate assays to assess inter-assay precision.
CV(%) = SD/mean x 100
Inter-Assay: CV< 10%
CV(%) = SD/mean x 100
Inter-Assay: CV< 10%
Intended Use
This Sandwich kit is for the accurate quantitative detection of Mouse Complement C1q tumor necrosis factor related protein 5 (also known as C1qtnf5) in serum, plasma, cell culture supernatants, Ascites, tissue homogenates or other biological fluids.
Assay Principle
This kit is an Enzyme-Linked Immunosorbent Assay (ELISA). The plate has been pre-coated with Mouse C1qtnf5 antibody. C1qtnf5 present in the sample is added and binds to antibodies coated on the wells. And then biotinylated Mouse C1qtnf5 Antibody is added and binds to C1qtnf5 in the sample. Then Streptavidin-HRP is added and binds to the Biotinylated C1qtnf5 antibody. After incubation unbound Streptavidin-HRP is washed away during a washing step. Substrate solution is then added and color develops in proportion to the amount of Mouse C1qtnf5. The reaction is terminated by addition of acidic stop solution and absorbance is measured at 450 nm.
| Reagent Provided | ||
| Components | Quantity (96T) | Quantity (48T) |
| Standard solution (16000ng/L) | 0.5ml x1 | 0.5ml x1 |
| Pre-coated ELISA plate | 12 * 8 well strips x1 | 12 * 4 well strips x1 |
| Standard diluent | 3ml x1 | 3ml x1 |
| Streptavidin-HRP | 6ml x1 | 3ml x1 |
| Stop solution | 6ml x1 | 3ml x1 |
| Substrate solution A | 6ml x1 | 3ml x1 |
| Substrate solution B | 6ml x1 | 3ml x1 |
| Wash buffer Concentrate (25x) | 20ml x1 | 20ml x1 |
| Biotinylated Mouse C1qtnf5 antibody | 1ml x1 | 1ml x1 |
| User instruction | 1 | 1 |
| Plate sealer | 2 pics | 2 pics |
Material Required but Not Supplied
- 37°C±0.5°C incubator
- Absorbent paper
- Precision pipettes and disposable pipette tips
- Clean tubes
- Deionized or distilled water
- Microplate reader with 450 ± 10nm wavelength filter
Precautions
- Prior to use, the kit and sample should be warmed naturally to room temperature 30 minutes.
- This instruction must be strictly followed in the experiment.
- Once the desired number of strips has been removed, immediately reseal the bag to protect the remain from deterioration. Cover all reagents when not in use.
- Make sure pipetting order and rate of addition from well-to-well when pipetting reagents.
- Pipette tips and plate sealer in hand should be clean and disposable to avoid cross-contamination.
- Avoid using the reagents from different batches together.
- Substrate solution B is sensitive to light, don’t expose substrate solution B to light for a long time.
- Stop solution contains acid. Please wear eye, hand and skin protection when using this material. Avoid contact of skin or mucous membranes with kit reagent.
- The kit should not be used beyond the expiration date.
Specimen Collection
Serum Allow serum to clot for 10-20 minutes at room temperature. Centrifuge at 2000-3000 RPM for 20 minutes. Collect the supernatant without sediment.
Plasma Collect plasma using EDTA or heparin as an anticoagulant. After mix 10-20 minutes, centrifuge samples for 20 minutes at 2000-3000 RPM. Collect the supernatant without sediment.
Cell culture supernatant Collect by sterile tubes. When detecting secrete components, centrifuge at 2000-3000 RPM for 20 minutes. Collect the supernatants. When detecting the components in the cell, use PBS (pH 7.2-7.4) to dilute cell suspension, the cell concentration of approximately 1 million/ml. Damage cells through repeated freeze-thaw cycles to let out the inside components. Centrifuge at 2000-3000 RPM for 20 minutes. Collect the supernatant without sediment.
Tissue Rinse tissues in ice-cold PBS (pH 7.4) to remove excess blood thoroughly and weigh before homogenization. Mince tissues and homogenize them in PBS (tissue weight (g): PBS (mL) volume=1:9) with a glass homogenizer on ice. To further break down the cells, you can sonicate the suspension with an ultrasonic cell disrupter or subject it to freeze-thaw cycles. The homogenates are then centrifuged for 15 minutes at 12,000 RPM at 4°C to get the supernatant. Avoid freeze/thaw cycles.
Urine/Ascites/Cerebrospinal fluid Collect by sterile tube. Centrifuge at 2000-3000 RPM for 20 minutes. Collect the supernatant without sediment.
Plasma Collect plasma using EDTA or heparin as an anticoagulant. After mix 10-20 minutes, centrifuge samples for 20 minutes at 2000-3000 RPM. Collect the supernatant without sediment.
Cell culture supernatant Collect by sterile tubes. When detecting secrete components, centrifuge at 2000-3000 RPM for 20 minutes. Collect the supernatants. When detecting the components in the cell, use PBS (pH 7.2-7.4) to dilute cell suspension, the cell concentration of approximately 1 million/ml. Damage cells through repeated freeze-thaw cycles to let out the inside components. Centrifuge at 2000-3000 RPM for 20 minutes. Collect the supernatant without sediment.
Tissue Rinse tissues in ice-cold PBS (pH 7.4) to remove excess blood thoroughly and weigh before homogenization. Mince tissues and homogenize them in PBS (tissue weight (g): PBS (mL) volume=1:9) with a glass homogenizer on ice. To further break down the cells, you can sonicate the suspension with an ultrasonic cell disrupter or subject it to freeze-thaw cycles. The homogenates are then centrifuged for 15 minutes at 12,000 RPM at 4°C to get the supernatant. Avoid freeze/thaw cycles.
Urine/Ascites/Cerebrospinal fluid Collect by sterile tube. Centrifuge at 2000-3000 RPM for 20 minutes. Collect the supernatant without sediment.
Note
- Sample concentrations should be predicted before being used in the assay. If the sample concentration is not within the range of the standard curve, users must contact us to determine the optimal sample for user’s particular experiment.
- Samples to be used within 5 days should be stored at 2-8°C. Samples should be aliquoted or must be stored at -20°C within 1 month or -80°C within 3 months. Avoid repeated freeze thaw cycles.
- Samples should be brought to room temperature before starting the assay.
- Centrifuge to collect sample before use.
- Samples containing NaN3 can’t be tested as it inhibits the activity of Horse Radish Peroxidase (HRP).
- Collect the supernatants carefully. When sediments occurred during storage, centrifugation should be performed again.
- Hemolysis can greatly impact the validity of test results. Take care to minimize hemolysis.
*Sample can't be diluted with this kit. Owing to the material we use to prepare the kit, the sample matrix interference may falsely depress the specificity and accuracy of the assay.
Reagent Preparation
- All reagents should be brought to room temperature before use.
-
Standard Reconstitute the 120ul of the standard (16000ng/L) with 120ul of standard diluent to generate a 8000ng/Lstandard stock solution. Allow the standard to sit for 15 mins with gentle agitation prior to making dilutions. Prepare duplicate standard points by serially diluting the standard stock solution (8000ng/L) 1:2 with standard diluent to produce 4000ng/L, 2000ng/L, 1000ng/L and 500ng/L solutions. Any remaining solution should be frozen at -20°C and used within one month. Dilution of standard solutions suggested are as follows:
- Wash Buffer Dilute 20ml of Wash Buffer Concentrate 25x into deionized or distilled water to yield 500 ml of 1x Wash Buffer. If crystals have formed in the concentrate, mix gently until the crystals have completely dissolved.
| 8000ng/L | Standard No.5 | 120ul Original standard + 120ul Standard diluent |
| 4000ng/L | Standard No.4 | 120ul Standard No.5 + 120ul Standard diluent |
| 2000ng/L | Standard No.3 | 120ul Standard No.4 + 120ul Standard diluent |
| 1000ng/L | Standard No.2 | 120ul Standard No.3 + 120ul Standard diluent |
| 500ng/L | Standard No.1 | 120ul Standard No.2 + 120ul Standard diluent |
| Standard concentration | Standard No.5 | Standard No.4 | Standard No.3 | Standard No.2 | Standard No.1 |
| 16000ng/L | 8000ng/L | 4000ng/L | 2000ng/L | 1000ng/L | 500ng/L |
Assay Procedure
- Prepare all reagents, standard solutions and samples as instructed. Bring all reagents to room temperature before use. The assay is performed at room temperature.
- Determine the number of strips required for the assay. Insert the strips in the frames for use. The unused strips should be stored at 2-8°C.
- Add 50ul standard to standard well. Note: Don’t add antibody to standard well because the standard solution contains biotinylated antibody.
- Add 40ul sample to sample wells and then add 10ul Mouse C1qtnf5 antibody to sample wells, then add 50ul streptavidin-HRP to sample wells and standard wells (Not blank control well). Mix well. Cover the plate with a sealer. Incubate 60 minutes at 37°C.
- Remove the sealer and wash the plate 5 times with wash buffer. Soak wells with 300ul wash buffer for 30 seconds to 1 minute for each wash. For automated washing, aspirate or decant each well and wash 5 times with wash buffer. Blot the plate onto paper towels or other absorbent material.
- Add 50ul substrate solution A to each well and then add 50ul substrate solution B to each well. Incubate plate covered with a new sealer for 10 minutes at 37°C in the dark.
- Add 50ul Stop Solution to each well, the blue color will change into yellow immediately.
- Determine the optical density (OD value) of each well immediately using a microplate reader set to 450 nm within 10 minutes after adding the stop solution.
Summary
- Prepare all reagents, samples and standards.
- Add sample and ELISA reagent into each well. Incubate for 1 hour at 37°C.
- Wash the plate 5 times.
- Add substrate solution A and B. Incubate for 10 minutes at 37°C.
- Add stop solution and color develops.
- Read the OD value within 10 minutes.
Calculation of Result
Construct a standard curve by plotting the average OD for each standard on the vertical (Y) axis against the concentration on the horizontal (X) axis and draw a best fit curve through the points on the graph. These calculations can be best performed with computer-based curve-fitting software and the best fit line can be determined by regression analysis.
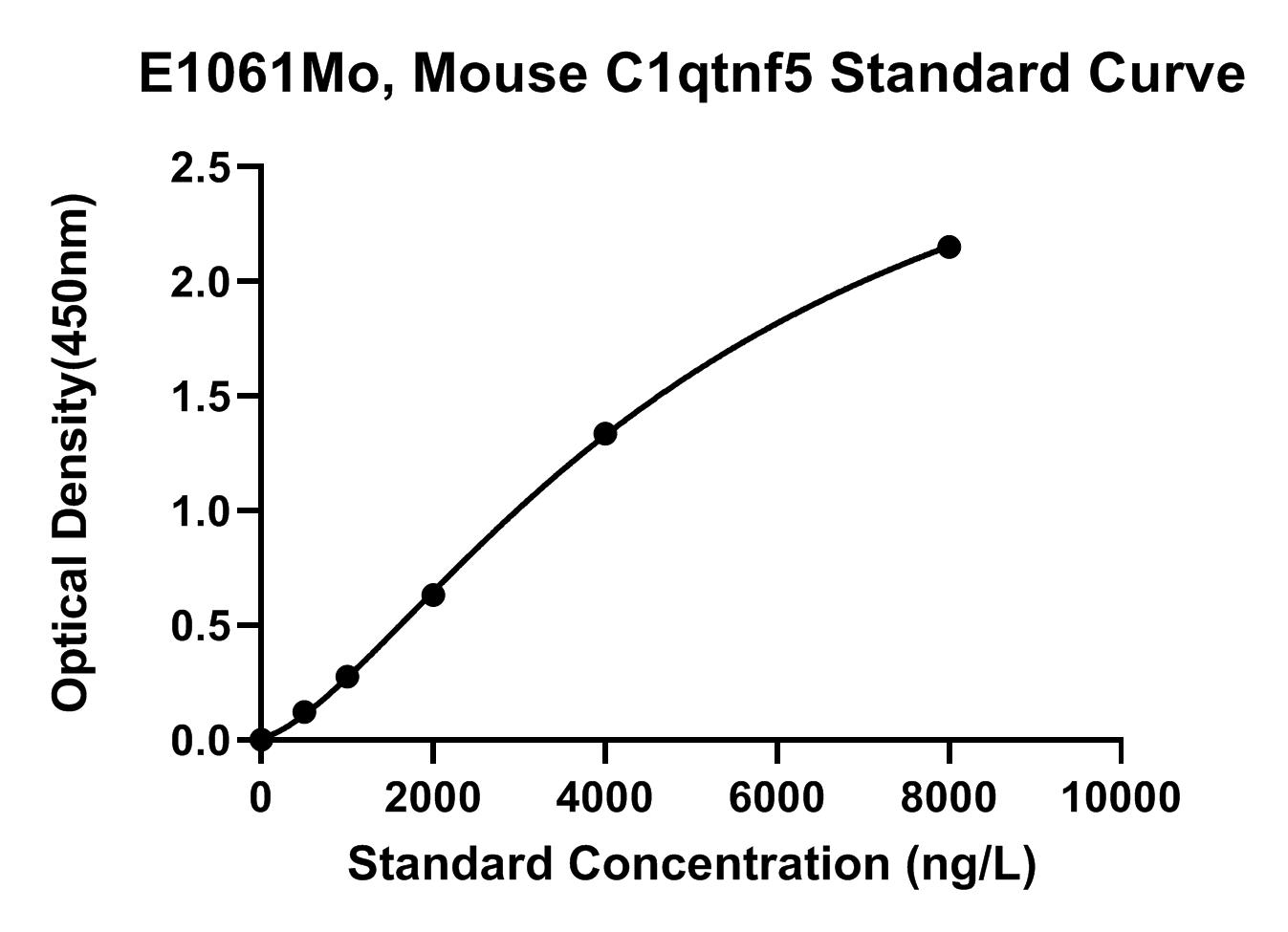
Typical Data
The standard curve of E1061Mo is provided for demonstration only. A standard curve should be generated for each set of samples assayed.
 |
||||||||||||||||||||||||||||
|
| Troubleshooting | |
| High Background possible case | Solution |
| Improper washing | Increasing duration of soaking steps |
| Incorrect incubation temperature | Incubate at 37°C |
| Incubation time too long | Reduce incubation time |
| Substrate exposed to light prior to use | Keep substrate in a dark place |
| Substrate was contaminated | Replace substrate. Substrate should be clean and avoid crossed contamination by using the sealer |
| Contaminated wash buffer | Use a clean buffers and sterile filter |
| Weak or No Signal possible case | Solution |
| A reagent or a step of the procedure omitted by mistake | Check protocol and follow steps carefully |
| Antibody are not enough | Increase the concentration of the antibody |
| Improper washing | Increasing duration of soaking steps |
| Reagent are contaminated | Use new one |
| Pipette are not clean | Pipette should be clean |
| HRP was not added | Add HRP according to the instruction |
| Sample contains sodium azide | Don't prepare samples with sodium azide |
| Wrong incubation time or temperature | Check and follow protocol. Place plates in an incubator during incubation periods (set to 37ºC). |
| Poor Precision possible case | Solution |
| Pipetting error | Dispense quickly and identically into the side of each well. Use calibrated pipettes. |
| Incomplete washing | Make sure wells are washed adequately by filling the wells with proper amount of wash buffer. |
| Unclean wells | Inspect wells and remove debris prior to use. Wipe the outer bottom of plate clean to remove any debris or fingerprints prior to reading. |
| Poor standard curve possible case | Solution |
| Incorrect preparation of standard | Reconstitute standard as suggested on data sheet. |
| Capture Antibody did not bind | Use BT LAB ELISA plate in the kit |
| Inefficient washing | Be sure wash apparatus is working properly (i.e. distributing even volumes into each well). Be sure wells are empty after aspiration, yet be sure to fill wells in a timely manner. |
| Pipetting error | Dispense quickly and identically into the side of each well. Use calibrated pipettes. |
| Incorrect storage of components | Store all components as recommended on data sheet. Do not allow reconstituted reagents to stay at room temperature for excess time. |
| If you have any question on the order please contact us via: [email protected]; technical assistance please contact us via: [email protected] More product visit www.bt-laboratory.com |